Imagine if curing genetic blindness was as simple as taking antibiotics, much like treating an infection with penicillin. Recent research from this year proposed the possibility of such a revolutionary approach to treatment of retinal disease, formerly known to be incurable.4
Traditionally, the human eye had been considered a sterile environment in medical research, protected and free from bacteria and other microbes. However, a study performed in 2021 turned this understanding on its head, demonstrating, through the collection and analysis of 1,000 human eye samples, the existence of a so-called “intraocular microbiome”, contained within the intraocular cavity of the eye.3 Along with this was the discovery of “disease-specific microbial signature[s]” associated with different eye diseases.3
A similar discovery was found for retinal degenerative disease, which causes retinal lesions that lead to blindness. Disease associated microbes, specifically “intralesional bacteria” (bacteria in retinal lesions, which are not normally found in the eye) were found in mice with CRB1 mutations, which are genetic mutations associated with retinal disease in both humans and mice.4
CRB1 mutations are associated with retinal degenerative diseases (also known as retinal dystrophies), such as Leber congenital amaurosis (LCA) and retinitis pigmentosa (RP) (which alone affects 5.5 million people worldwide).4,5 Common symptoms of these diseases (in children and young adults) include color blindness, night blindness, peripheral vision blindness, and, in some cases, complete blindness.2
CRB1 mutations were found to impair the “outer blood retinal barrier,” impair the colonic intestinal epithelial barrier, and cause retinal lesions in mice.4,1 This thinning of the barrier of the retina (similarly found in human patients with this mutation) allowed for the degradation of the retina, as well as the translocation of bacteria from the intestinal microbiome to the retina. When the bacteria was able to reach the thinned wall of the retina from the gut, it most likely caused lesions (or small cuts) at the retinal barrier. This resulted in retinal degradation in the CRB1 mutation mice.4 Thus, it seemed plausible to researchers that antimicrobial agents would have the ability to kill off the intestinal bacteria that was traveling to the retina, and thus prevent retinal degradation.
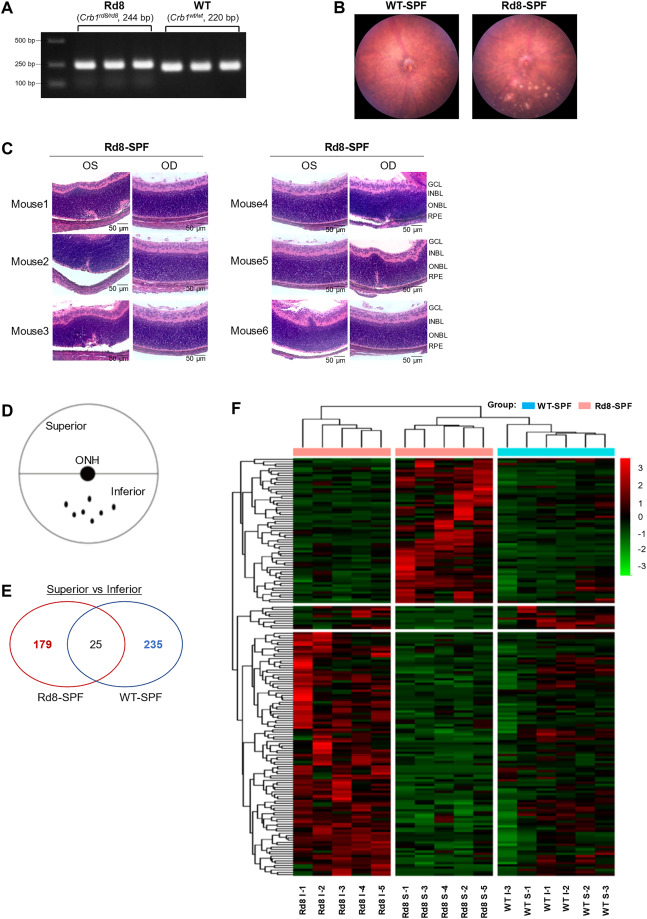
Figure 1: Mouse with wildtype (no CRB1 mutation) with Healthy Retina, Above right: Mouse with Rd8 mutation in the CRB1 gene, white spots in retina, characteristic of retinal degradation
Researchers administered treatments to mice with CRB1 mutations in three methods, all of which prevented the appearance of retinal lesions in the mice.4
First, CRB1 mutant mice (also known as Rd8 mice) were grown in germ-free conditions. This meant that the mice were grown in a completely sterile environment, having access to no microbes, and thus never developing a microbiome. These Rd8 mice showed no sign of retinal lesions, although the outer barrier of the retina showed a slight disruption due to the mutation they carried. Furthermore, when these germ-free mice were raised in an SPF environment (where some microbes are present, and thus the mice form a microbiome), retinal lesions began to appear in the Rd8 mice. This demonstrated that there was a significant relationship between bacteria and retinal lesions and Rd8 mice.
Second, a sort of vaccine, called an “adeno-associated virus (AAV) 2 viral vector system” was used to induce normal, non-mutated CRB1 protein in the intestine of Rd8 mice. This resulted in a stronger intestinal epithelial barrier, and finally in significantly reduced lesions in the Rd8 mice. Thus, controlling the expression of the CRB1 gene in the gut resulted in reduced retinal degeneration.
Third, a broad-spectrum antibiotic cocktail was administered to pregnant Rd8 mice living in a non-sterile environment. The Rd8 pups continued to be treated with these antibiotics before and after weaning, and showed fewer retinal lesions of smaller size as compared to Rd8 mice without antibiotics. Thus, antibiotics could be used as a treatment in the future to prevent the progression of retinal degradation.
Along with these methods are of course drawbacks, the study was conducted on neonatal mice, which can be significantly different when translating to human patients, especially patients that are older (such as children and young adults that are most affected by this disease). Another concern may be the development of antibiotic resistant bacteria in response to antibiotics administered to stop the disease.
However, taken together, these three findings show the critical role of gut bacteria in retinal degeneration and a possible solution to treating the disease, which may be as simple as taking antibiotics. The findings shed hope on finding treatments for formerly “incurable” retinal diseases and provide a path forward for other common eye diseases such as glaucoma and age related macular degeneration which have also been associated with microbial signatures.3
More broadly, findings such as these show the importance and power of challenging long-standing beliefs and assumptions (in this case, the sterility of the eye) in order to progress forward in research and, ultimately, find treatments and solutions for those who need it most.
References
- Beryozkin A., Zelinger L., Bandah-Rozenfeld D., Harel A., Strom T.A., Merin S., Chowers I., Banin E., Sharon D. (2013, March 1). Mutations in CRB1 Are a Relatively Common Cause of Autosomal Recessive Early-Onset Retinal Degeneration in the Israeli and Palestinian Populations. Investigative Ophthalmology & Visual Science.https://iovs.arvojournals.org/article.aspx?articleid=2166180#:~:text=Mutations%20in%20Crumbs%20homolog%201,to%20retinitis%20pigmentosa%20(RP).
- Chawla, H. (2023, March 16). Retinal Dystrophies. StatPearls [Internet] https://www.ncbi.nlm.nih.gov/books/NBK564379/.
- Deng, Y., Ge, X., Li, Y. et al. (2021, January 26). Identification of an intraocular microbiota. Cell Discovery. Nature. https://doi.org/10.1038/s41421-021-00245-6
- Peng S., Li J.J., Song W., Li Y., Zeng L., Liang Q., Wen X., Shang H., Liu K., Peng P., Xue W., Zou B., Yang L., Liang J., Zhang Z., Guo S., Chen T., Li W., Jin M., Xing X.B., Wan P., Liu C., Lin H., Wei H., Lee R.W.J., Zhang F., Wei L. (2024, February 26). CRB1-Associated Retinal Degeneration Is Dependent on Bacterial Translocation from the Gut. Cell. https://pubmed.ncbi.nlm.nih.gov/38412859/.
- Sidik S. (2024, February 26). The surprising link between gut bacteria and devastating eye Diseases. Nature News. https://www-nature-com.libproxy.berkeley.edu/articles/d41586-024-00562-2
