The Push for Biotechnology
It is difficult to pinpoint when “biotechnology” began, but many would agree that this industry will revolutionize our future. The term was first coined in 1917 by Karl Ereky to describe parallels between microorganisms and machines.1 However, the biotechnology industry did not gain the attention of policymakers until the rising oil prices of the late 1970s called traditional energy practices into question.1 Fast forward to the early 20th century, the completion of the Human Genome Project was compared to the invention of the wheel by the Director of Britain’s Wellcome Trust. 1 Representing over 47 years of research and nearly 3 billion dollars (roughly 5 billion dollars today, adjusted for inflation),2 the Human Genome Project brought together twenty institutions to produce a genetic sequence that accounted for 90% of the human genome. This innovation set the stage for a new frontier in genetic research, and, perhaps more importantly, reflected a significant public interest in biotechnology.
As we stand at the cusp of a future enriched with innovations like CRISPR-Cas9, bioprinting, nanomedicine, and brain-computer interfaces, public sentiments oscillate between anticipation and apprehension. Given the transformative nature of these advancements, it’s crucial to delve into the legal framework that will safeguard and guide this technological revolution.
The Importance of Patents in Biotechnology
Understanding biotechnology’s ties to patents begins with a grasp of the patent system’s core principles. The United States Patent and Trademark Office (PTO) has a role laid out by Article 1, Section 8, Clause 8 of the U.S. Constitution “to promote the Progress of Science and useful Arts.”15 The PTO attempts to fulfill this duty by creating “effective mechanisms that protect new ideas and investments in innovation and creativity.”18 The PTO issues ‘patents,” which grant the holder the right to exclude others from using the invention described in the patent. A patent asks an inventor to lay bare the inner workings of their invention to the public. In exchange, the government will grant the inventor exclusive rights over the manufacturing, selling, and licensing of their product for twenty years.
However, a few criteria must be met before an invention is deemed patentable. Namely, the invention must be novel, possess utility, and be non-obvious.4 In the context of biotechnology, it is important to note that since patents protect inventions, under current case law scientific discoveries themselves are not patentable. In other words, for something to be patentable, it must be created, rather than discovered.
The average cost to bring a new therapeutic to market is 1.3 billion dollars.5 Both the high cost of research and development and the long lead times required to develop a new therapeutic are thought to justify the use of patents in biotechnology. Companies rely on patents to protect their temporal and monetary investments in research and development. If another firm is authorized to reverse-engineer its competitor’s breakthrough, then a biotechnology firm has no reason to adopt the steep investment of bringing a product to market.

Even in academia, patents are extremely prevalent as they signify the originator of a process or invention and are a huge revenue generator for universities. Currently, UC Berkeley possesses 16 patents surrounding Jennifer Doudna’s groundbreaking CRISPR-Cas9 technology.6 Such a portfolio will likely bring the university anywhere from 100 million to 10 billion dollars over the next two decades.7 Such patents are an integral and necessary part of almost any invention in biotechnology.
The History of Biotechnology and Patent Protection
A history of friction exists between the patent system and biotechnology. However, this may not be a surprise. The USPTO is a federal agency established in the 19th century that is attempting to regulate new and upcoming technology that would have been unfathomable to its founders.
The first friction point between biotechnology and patents stems from the fact that biological research often includes isolating products of nature. In 1900, the pharmaceutical company, Parke-Davis & Co. patented epinephrine due to its lucrative potential.8 Shortly after, the patent was challenged by H. K. Mulford Co. on the basis that the compound was present in all humans, and thus no novel product was created. Nonetheless, the court concluded that the isolated and purified epinephrine was “for every practical purpose a new thing commercially and therapeutically.”9 Thus, a precarious precedent was established: nature’s wonders, once isolated, were seemingly novel inventions.
Secondly, scientists have long been wary of granting patents for living organisms. This issue was raised in 1972 when Ananda Chakrabarty was denied a patent on a recombinant bacterium. The PTO swiftly declined Chakrabarty’s patent application, deeming living entities beyond the realm of patentability.10 Eventually, the parties in Diamond v. Chakrabarty found themselves before the Supreme Court where the majority ruled in favor of Chakrabarty, famously stating that “anything under the sun that is made by man” could indeed be patented.11
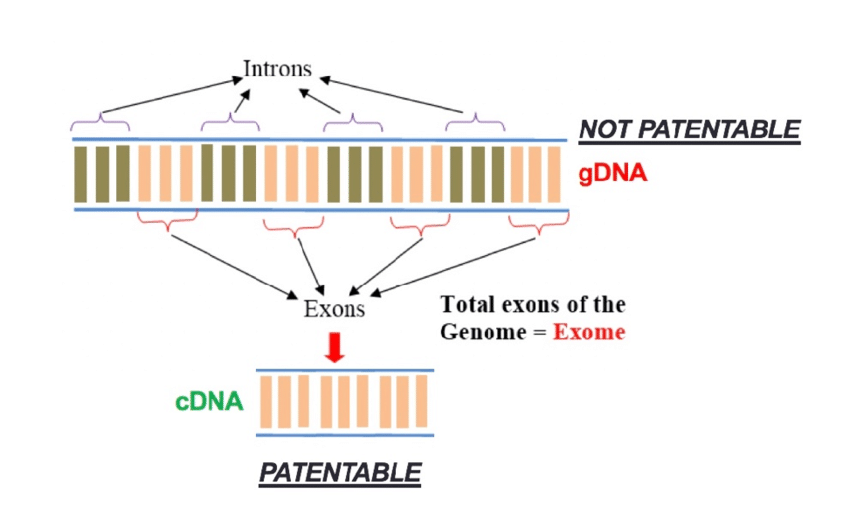
Both the Parke-Davis and Diamond v. Chakrabarty decisions laid the groundwork for the thousands of genome patents that were issued in the ‘80s, ‘90s, and ‘00s. However, in 2013, over 4,000 gene patents were deemed invalid due to Myriad v. Association of Molecular Pathology (AMP) which concluded that genes were unpatentable as they were merely isolated products of nature.12 This decision completely overturned the precedent set by Parke-Davis. Yet, in this same decision, complementary DNA (cDNA) was deemed patentable as it was “unnatural.” However, cDNA is simply genetic DNA without the portions that do not code for proteins (introns).
As a result, cDNA is, in many cases, a more useful form of DNA for an array of laboratory procedures. Thus, the Myriad v. AMP decision, in effect, does not prevent companies from gaining exclusive rights over portions of the genome, but this “loophole” was no secret. A 2015 report concludes that “there are many different ways to ‘claim around’ Myriad, allowing companies and universities to fully protect their DNA-based inventions”.14 This highlights one major underlying issue between the patent system and biotechnology; the rules for patent eligibility are set by nonscientists who are not best equipped to understand the ramifications of their policy.
Modernizing the U.S. Patent and Trademark Office
The tension between the USPTO and biotechnology is apparent, and with each judicial ruling, the confidence of biotechnology companies in the system meant to safeguard their innovations wanes. To bring the patent system into the 21st century—a system that has seen little change over the past 180 years—the USPTO should be granted substantive rulemaking authority.
At present, the criteria for what is patentable are subject to change only through legislative action by Congress or through changing interpretations by the Supreme Court. However, “since 1952 Congress has not taken much interest in amending the patent code leaving the bulk of legal evolution to the courts.”16 This, as previously highlighted, has led to changes in the patentability criteria only occurring after costly cases make their way to the Supreme Court. By granting the PTO substantive rulemaking authority, the agency would be empowered to define patent standards grounded in up-to-date science. This would enable a nimble, yet appropriate adaptation of rules to keep pace with rapid advancements in biotechnology.13
While this suggestion may seem autocratic, such autonomy would help the PTO’s regulations align with evolving industry standards. Additionally, this type of authority is not uncommon; agencies such as the Food and Drug Administration (FDA), the Federal Trade Commission, and the U.S. Securities and Exchange Commission already independently promulgate policy within their respective domains.13 As well, in each example of substantive rulemaking authority, any rule deemed “unreasonable” can be invalidated by the Supreme Court.13
As illustrated by Rochelle Dreyfuss, “[The PTO’s] expertise as scientists and their experience as examiners would sharpen interpretation of factual matters dispositive of patent quality […] Furthermore, their caseload would position them to make adjustments as they see how the rules play out (for example, how claim scope affects claiming strategies).”17 In essence, substantive rulemaking authority would transfer the responsibility of setting patentability criteria—a largely scientific endeavor—from the hands of non-experts to industry specialists. Undoubtedly, if the PTO gained the authority to set the criteria for patentability, shifts in funding and oversight would be necessary to ensure the PTO remains unbiased and uninfluenced by monetary gain. Nonetheless, granting the PTO substantive rule-making authority should be a topic that enters the public conversation.
Certainly, no expert can foresee all future conflicts between science and law and disputes will inevitably continue to escalate to the Supreme Court for review. However, what the public understands currently about biotechnology is merely the tip of the iceberg relative to the industry’s potential. Given the profound impact biotechnology is poised to have on our future, it is prudent to establish a patent system that is equipped to respond to the industry’s ever-changing and complex advancements.
Acknowledgments
I would like to express my sincerest gratitude to Professor Schmitt, a professor at UC Berkeley School of Law who, in addition to possessing a JD, holds a PhD in Physical Biochemistry. Professor Schmitt provided detailed feedback to ensure accuracy and clarity.
References
1. Bud, R. (2003). History of Biotechnology. In Encyclopedia of Life Sciences. John Wiley & Sons, Ltd. https://doi.org/10.1038/npg.els.0003086 Link
2. Human Genome Project Fact Sheet. (n.d.). Genome.Gov. Retrieved October 27, 2023, from https://www.genome.gov/about-genomics/educational-resources/fact-sheets/human-genome-project Link
3. Alexander Fleming Discovery and Development of Penicillin—Landmark. (n.d.). American Chemical Society. Retrieved October 27, 2023, from https://www.acs.org/education/whatischemistry/landmarks/flemingpenicillin.html Link
4. The Natural Complexity of Patent Eligibility | Iowa Law Review—The University of Iowa. (n.d.). Retrieved October 27, 2023, from https://ilr.law.uiowa.edu/print/volume-99-issue-3/the-natural-complexity-of-patent-eligibility Link
5. Average cost of developing a new drug could be up to $1.5 billion less than pharmaceutical industry claims | LSHTM. (n.d.). Retrieved October 27, 2023, from https://www.lshtm.ac.uk/newsevents/news/2020/average-cost-developing-new-drug-could-be-15-billion-less-pharmaceutical Link
6. UC now holds largest CRISPR-Cas9 patent portfolio. (2022). Berkeley. https://news.berkeley.edu/2019/10/01/uc-now-holds-largest-crispr-cas9-patent-portfolio Link
7. Complex CRISPR Patent Decision Benefits Broad Institute, Editas. (n.d.). BioSpace. Retrieved October 27, 2023, from https://www.biospace.com/article/latest-crispr-patent-battle-benefits-broad-institute-of-harvard-and-mit-/ Link
8. Beauchamp, C. (n.d.). Patenting Nature: A Problem of History. 16. Link
9. Parke-Davis & Co. v. H.K. Mulford. 1911. 189 F. 95 (S.D.N.Y.) Link
10. Czarnetzky, J. M. (1988). Altering Nature’s Blueprints for Profit: Patenting Multicellular Animals. Virginia Law Review, 74(7), 1327. https://doi.org/10.2307/1073274 Link
11. Diamond v. Chakrabarty. (n.d.). Oyez. Retrieved October 27, 2023, from https://www.oyez.org/cases/1979/79-136 Link
12. Klein, R. D. (2013). AMP v Myriad: The Supreme Court Gives a Win to Personalized Medicine. The Journal of Molecular Diagnostics, 15(6), 731–732. https://doi.org/10.1016/j.jmoldx.2013.09.002 Link
13. Barfield, C. E., & Calfee, J. E. (2007a). Biotechnology and the patent system: Balancing innovation and property rights. Published for the American Enterprise Institute [by] AEI Press. Link
14. Wales, M., & Cartier, E. (2015). The Impact of Myriad on the Future Development and Commercialization of DNA-Based Therapies and Diagnostics. Cold Spring Harbor Perspectives in Medicine, 5(12), a020925. https://doi.org/10.1101/cshperspect.a020925 Link
15. U.S. Const. Art. 1, § 8, Clause 3.
16. Long, C. (2009). The PTO and the Market for Influence in Patent Law. University of Pennsylvania Law Review, 157(6). Link
17. Dreyfuss, R. (2006). Pathological Patenting: The PTO as Cause or Cure . Michigan Law Review, 104(6). Link
18. About Us. United States Patent and Trademark Office – An Agency of the Department of Commerce. (2022, November 7). Link
