Your gums do not regenerate after gum disease—it is impossible. For people who don’t brush and floss two times a day, periodontitis (advanced gum disease) quickly forms. Periodontitis is caused by the inflammation and infection of tissues and bone that support the teeth, including the cementum, gingiva, periodontal ligament, and alveolar bone.
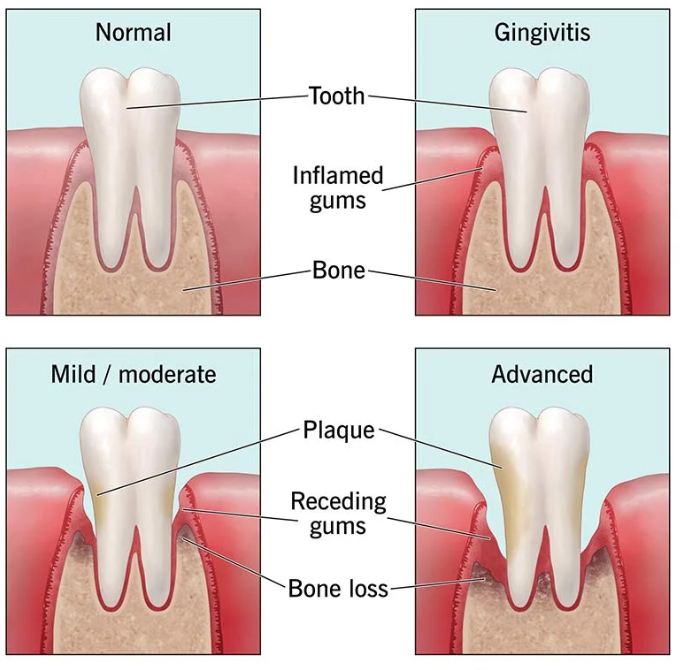
Without proper intervention, this disease can cause gum recession, tooth loss, and the destruction of the jawbone. Currently, 47% of all adults 30 years or older in the United States suffer with this disease and experience tissue loss as a result.1
Current Treatments for Periodontitis
If someone with periodontitis were to walk into a dentist’s office for treatment, they would be given two main options: gum and bone grafting, or root planing.
Gum and bone grafting involves taking gum and bone material from either one’s own body or a donor and surgically inserting this material into the periodontal cavity to provide support to the teeth and reduce mobility that’s caused by loss of gingiva and alveolar bone.
Although grafting can relieve the effects of periodontitis and prevent further damage by reinforcing the teeth, it does not reverse the damage caused by the disease since there is no stimulated tissue growth. As this is a surgical procedure, there can be other complications that arise with grafting. Oftentimes, there is a chance that the body will reject the graft tissue, which ultimately leads it to turn white and die. Additionally, grafting usually has a recovery period of around two weeks to two months, throughout which the patient may experience increased pain and discomfort.1
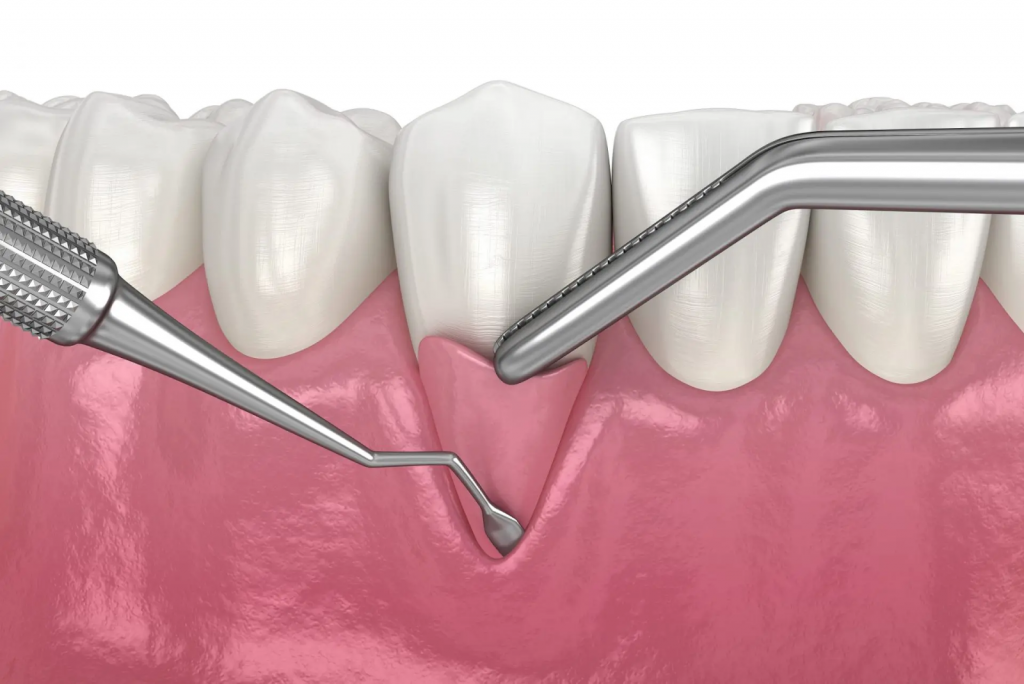
If the patient decides to opt out of bone and gum grafting they could also get a root planing, which is a slightly less invasive non-surgical procedure. Periodontitis develops a gap between the gum and the root where the gum tissue pulls away from the teeth due to inflammation. Root planing cleans the surface of the tooth so that the tissue can reattach and further damage is prevented.1 This method, similar to the grafting technique, can not reverse damage, and can only salvage what is left. These shortcomings of current treatment options have led to novel innovations at the intersection of dentistry and regenerative medicine, where researchers hope to find a way to not only protect and preserve healthy gum tissue, but also regenerate and replace the diseased tissue found in periodontitis patients.
The Use of Nanomaterials in Dentistry
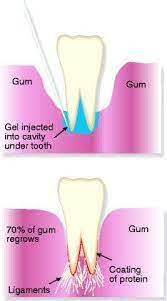
A possible answer lies in nanomaterials, chemical substances formed from incredibly small particles. This is an alternative to traditional clinical approaches that can result in regenerated tissue and long term success for treating periodontitis. Currently in clinical practice, though not widely adopted, this treatment involves the use of tissue-stimulating proteins in the gingival cavity, the space in between the tooth and the gingiva, or gum. Amelogenin (a protein found in infants’ mouths during teeth growth) is formulated in a hydrogel scaffold (a 3D framework that can hold large amounts of liquid) and injected into the affected area.2
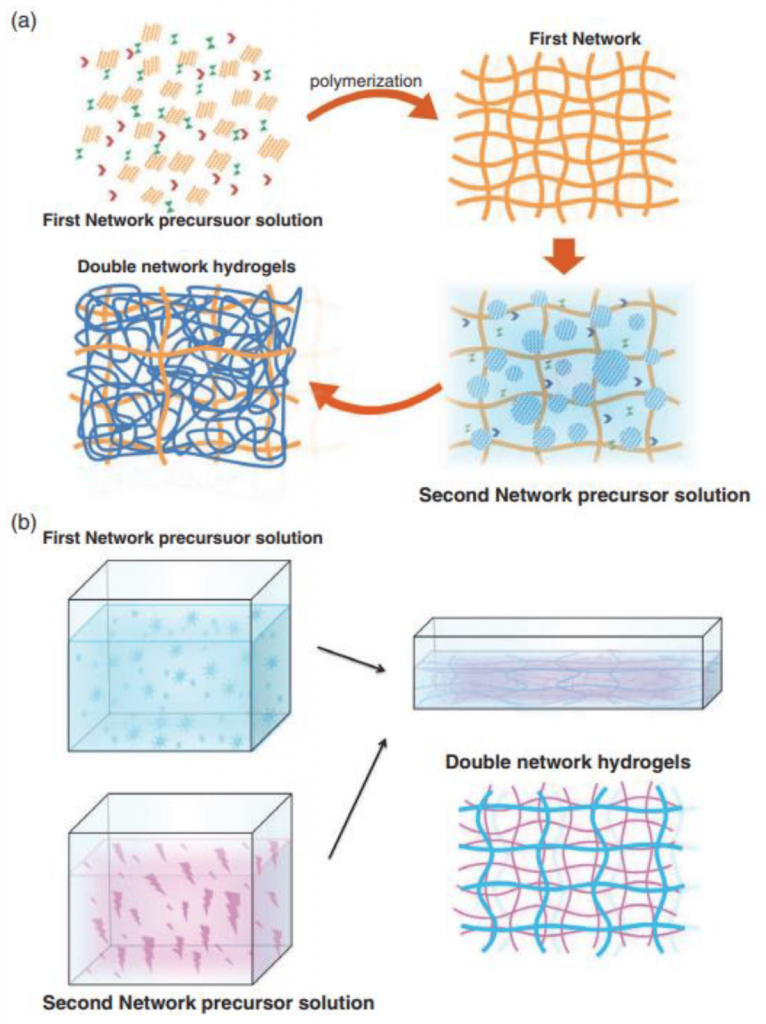
The protein coats the surface of the root and interacts with cells such as cementoblasts, which are found within the tissue. Cementoblasts are cells responsible for the generation of hard tissue found in between the tooth and gingival tissue called cementum. Due to its prominence during tooth development, the presence of amelogenin signals the cementoblasts to start producing tissue, resulting in gum regrowth.3
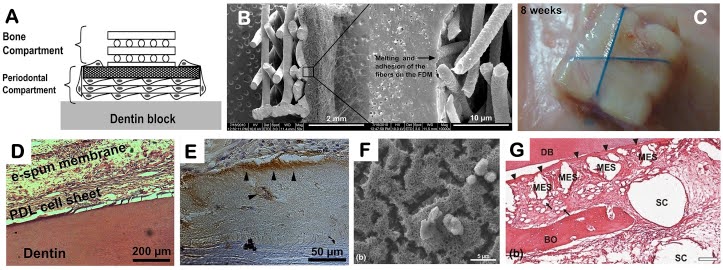
Another type of scaffolding used to potentially regenerate tissue are nanofibrous scaffolds, which simulate the extracellular matrix of gingival tissue, an environment where cells and proteins interact. One category of proteins, called growth factors, are integral to this process.4
Nanofibrous scaffolds carry these growth factors, specifically the ones that stimulate alveolar bone, cementum, and gingiva growth. When inserted into a gingival cavity, the scaffolding is adopted by nearby tissue and the growth factors spur a chain reaction of tissue growth.5
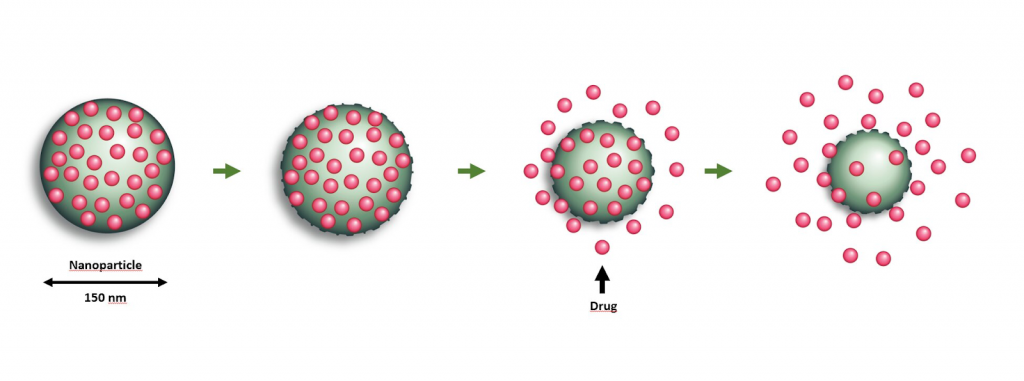
Outside of the use of scaffolds, researchers have been utilizing drug-carrying nanoparticles to encapsulate various drugs and antibiotics such as chitosan, as well as tetracycline and glycolic acids, which both have antibacterial properties, to reduce inflammation of the periodontal tissue and promote regeneration.
Chitosan is a polymer that inhibits the growth of mouth bacteria and also stimulates the activity of fibroblasts, a type of cell that generates connective tissue and maintains the framework of tissues during injury. By creating nanoparticles that can slowly release these drugs into periodontal pockets, there is a constant inflow of polymers to inhibit bacterial growth and consequent damage and regrow tissue. Researchers found that these nanoparticles have been able to regenerate bone and gum tissue in dogs; however, further research must be conducted before these novel nanoparticles may be used as a regular treatment option.6
Drawbacks of Nanomaterials
Despite the vast potential nanomaterials solutions have, there are also critical challenges associated with them to consider that may prevent immediate clinical use. Although nanomaterials can stimulate tissue growth, it is still uncertain how to regulate the ways in which the produced tissue grows back. As a result, there is possibility for tissue overgrowth, tissue disfiguration, or other changes in aesthetical properties compared to native tissue.1 Additionally, more intensive research and clinical trials will be necessary to fully understand any immunological reactions nanomaterials can induce in the long-term.
Future Implications of Nanomaterials
Currently, nanomaterial research within clinical use is only limited to hydrogel scaffolds. However, further research exploring fibrous scaffolding and drug nanoparticles can allow for more specialized options for those who are experiencing different degrees of periodontitis. People experiencing more issues with the infection aspect of periodontitis may prefer drug nanoparticles, while those more affected by tissue loss might prefer fibrous scaffolding. Additionally, by utilizing nanomaterials as a primary treatment to periodontitis, many drawbacks of more generalized treatments are resolved, including painful and long recovery times and the inability to restore tissue to its original state.
References
- professional, C. C. medical. (n.d.). Periodontal (gum) disease: How serious is it?. Cleveland Clinic. https://my.clevelandclinic.org/health/diseases/21482-gum-periodontal-disease
- Lee, B.-S., Lee, C.-C., Wang, Y.-P., Chen, H.-J., Lai, C.-H., Hsieh, W.-L., & Chen, Y.-W. (2016, January 18). Controlled-release of tetracycline and lovastatin by Poly(D,L-Lactide-co-glycolide acid)-chitosan nanoparticles enhances periodontal
- Liang, Y., Luan, X., & Liu, X. (2020, February 28). Recent advances in periodontal regeneration: A biomaterial perspective. Bioactive materials. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7052441/
- Zong, C., Bronckaers, A., Willems, G., He, H., & Cadenas de Llano-Pérula, M. (2023, May 24). Nanomaterials for periodontal tissue regeneration: Progress, challenges and future perspectives. MDPI. https://www.mdpi.com/2079-4983/14/6/290#B13-jfb-14-00290
- Nikoloudaki, G. (2021, June 21). Functions of matricellular proteins in dental tissues and their emerging roles in orofacial tissue development, maintenance, and disease. International journal of molecular sciences. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8235165/
- regeneration in dogs. International journal of nanomedicine. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4723100/