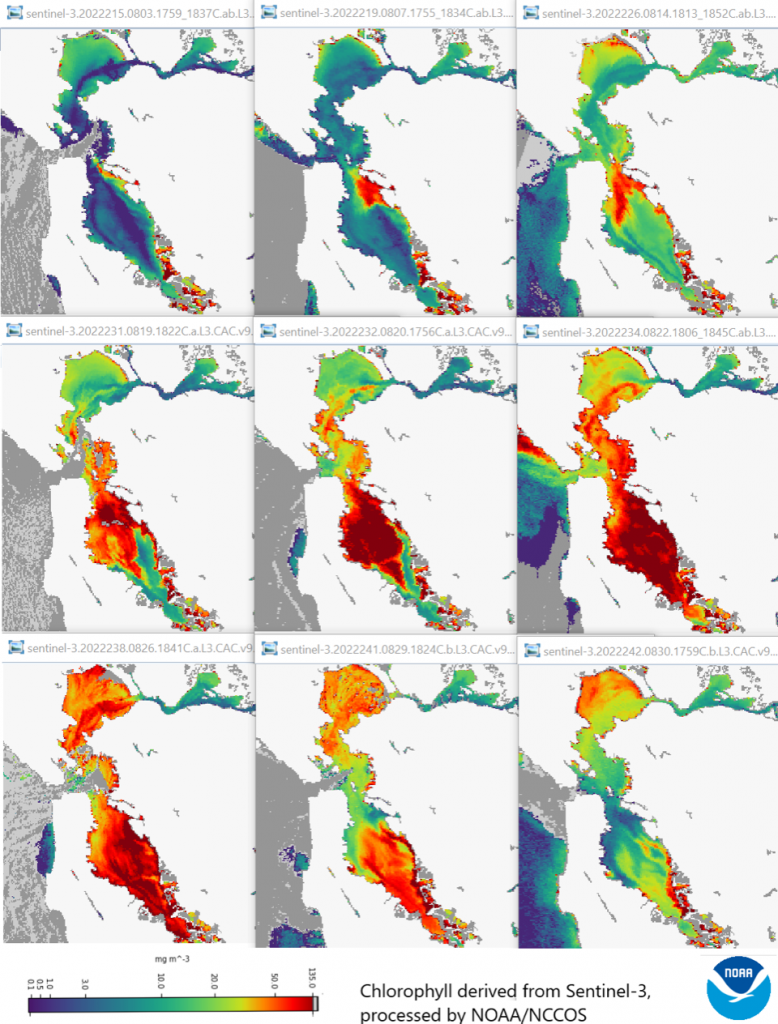
It’s July 2022, and the San Francisco Bay has turned a murky brown. Scattered reports of skin irritation and respiratory complications surface, followed by thousands of dead fish sweeping onto Bay Area shores.1
This is no freak die-off, though; San Francisco’s waterways are being choked by masses of Heterosigma akashiwo, a species of photosynthetic algae, in the most severe algal bloom the region has experienced to date. Months will pass before the bloom, sustained by warm waters polluted with excess nitrogen and phosphorus, finally subsides.2
In uncontrolled blooms like the one San Francisco experienced last summer, algae’s swift reproduction rate and ability to subsist on limited resources allow these unsightly algal aggregations to devastate ecological balance and threaten public health. On the other hand, these same traits also make algae a potent candidate for commercial application. Already, certain species of unicellular microalgae have found their market footholds as nutritional supplements, pharmaceuticals, and bioremediation agents in a rapidly expanding, multibillion-dollar industry.3
Among the most promising avenues of development for algae is in bioenergy. In fact, Heterosigma akashiwo itself has been cited for its potential to be converted from health hazard to viable source of biofuel.4
Biofuels differ from fossil fuels, such as coal and petroleum, in that they are derived directly from living, rather than long-dead, organic material. The interest in using living organisms, often fast-growing crops like corn, switchgrass, and soybean, lies mainly in the fact that these plants naturally reuptake the carbon dioxide that is released when fuel is burned. Those who back biofuels contend that this strategy of closing the “energy loop” will play a key role in cutting back on atmospheric carbon dioxide, a major greenhouse gas responsible for climate change and ultimately large-scale biodiversity loss.5 There’s just one problem: biofuel crops fail to match fossil fuels in energy production.
Hunting for Hydrocarbons
The energy deficit between bioenergy crops and fossil fuels is due to differences in chemical structure. The main output of growing bioenergy crops is lignocellulosic matter, a dry, fibrous plant material composed of lignin, cellulose, and hemicellulose, which are made up of carbon, hydrogen, and oxygen.6 Meanwhile, 97% of commercial petroleum consists of hydrocarbons with negligible amounts of oxygen.7
It’s this presence or absence of oxygen that plays a key role in determining the molecule’s energetic capacity. When fuels are combusted, high energy chemical bonds between hydrogen and carbon in the fuel source are broken down to release useful energy. As these bonds are broken, some atoms are reshuffled and new chemical bonds form. An important example of this is oxidation. This process occurs when fuel is combusted and ambient oxygen atoms are incorporated into the broken-down molecules to produce water, carbon dioxide, and other waste products.8
A fuel feedstock like lignocellulose that initially contains oxygen is already partially oxidized at the beginning of the reaction. For these fuels, less bonds need to be broken and rearranged to convert the fuel into its final product, meaning less energy is released from the reaction. As a result, compounds like lignocellulose with lower hydrogen to carbon ratios and carbon to oxygen ratios tend to produce fuels that are less dense in energy.9
Meanwhile, microalgae lack the lignocellulosic mass used in traditional biofuels and thus may offer a more efficient alternative. Algae yield greater proportions of high-energy compounds with higher hydrogen to oxygen ratios, particularly from its membranes and energy-storing lipids. Various species of algae are reported to contain anywhere from 20-50% lipids by weight,10 thus increasing the amount of extractable energy compared to its plant-based counterpart: the same acre that produces 18 gallons of oil from corn can yield up to 6,000 gallons of biodiesel if used to produce microalgae.11
These algal lipids are primarily triacylglycerols (TAGs) and diacylglycerols (DAGs). In a process known as transesterification, TAGs and DAGs react with small alcohols, such as methanol, to form long-chain fatty acid esters—the basis of biodiesel.12 Since most microalgae technology is not yet optimized to have our economy run exclusively on biodiesel, the current product can either be mixed into regular diesel to supplement petroleum-based fuel or used in its pure form in a limited number of diesel engines.13
The Dark Side of Sunlight Energy
In theory, microalgae offer a feasible and scalable source of energy. Algae do not need to be grown on arable land as crops do, and the Department of Energy estimates that reserving roughly 15,000 square miles for algae production, an area roughly the size of Maryland, would provide enough biomass to meet all petroleum demand in the United States.14
However, major inefficiencies in producing algal biofuels will have to be resolved before algae can support an entire country. While the appeal of algal biofuels is mainly due to its unique ability to turn sunlight—an unlimited and carbon-free resource—into fuel, solar energy is unavailable at nighttime and diffuse during periods of heavy cloud cover.
To make a bad problem worse, just 30% of incident sunlight that reaches the Earth can be absorbed by the plants and used in photosynthesis by the algal photosystems, which operate solely at the 680 and 700 nm, or red, wavelengths. After lipid extraction and processing, this number shrinks to a slim 8-10% theoretical maximum of sunlight’s original energy that is actually harnessed in biofuels.15
To mitigate these issues, methods of artificially accumulating TAGs, the primary energy storage lipid in algae, remain a major area of development. Much of algal research is in determining environmental conditions associated with optimal algal growth and lipid production. Currently, there is evidence that modulating everything from light intensity to carbon dioxide concentration, the length of light and dark cycles, and temperature may induce an increase in lipid metabolism. Another approach has been stressing algae, often by depriving them of nutrients or adding toxic levels of heavy metals or salts to the culture. Starving, toxin-stressed algae may invest more effort in storing energy in the form of TAGs. Still another strategy is to engineer genes that control algal metabolism to upregulate TAG production.16
A Future in Photosynthesis
As it stands, microalgae are largely overlooked and underutilized as a source of energy. Yet, San Francisco proved last summer that microalgae—by virtue of inborn hardiness and anthropogenic activity—are becoming an increasingly abundant resource, with or without the development of lab-raised strains. At the same time, dire consequences of climate change call more urgently than ever for a reliable source of sustainable energy, and growing evidence supports both bioprospecting naturally occurring algae and engineering algae in the lab as viable energy production strategies.17 If it’s greener energy that the planet needs to embrace, the solution (both in hue and function) may very well have been blooming in our periphery all along.
Acknowledgements
I would like to thank Dr. Anastasios Melis, a professor in the Department of Plant and Microbial Biology at UC Berkeley, both for introducing me to microalgal biofuels in his class and providing invaluable advice, expertise, and feedback in writing in this article. I would also like to thank Varun Upadhyay, the Berkeley Scientific Journal Features editor, for his guidance and support.
References
- NCCOS Assists Response to Harmful Algal Bloom in the San Francisco Bay. (2019, July 11). NCCOS Coastal Science Website. https://coastalscience.noaa.gov/news/nccos-assists-response-to-harmful-algal-bloom-in-the-san-francisco-bay/
- Harmful Algal Bloom in San Francisco Bay Results in Aquatic Mortality, Fish Kills – California Ocean Protection Council. (n.d.). Retrieved April 1, 2023, from https://www.opc.ca.gov/2022/09/harmful-algal-bloom/
- Phang, S.-M. (2010). Potential Products from Tropical Algae and Seaweeds, especially with Reference to Malaysia. Malaysian Journal of Science, 29(2), 160–166. https://doi.org/10.22452/mjs.vol29no2.7
- Stewart, J. J., Bianco, C. M., Miller, K. R., & Coyne, K. J. (2015). The Marine Microalga, Heterosigma akashiwo, Converts Industrial Waste Gases into Valuable Biomass. Frontiers in Energy Research, 3. https://doi.org/10.3389/fenrg.2015.00012
- Razzak, S. A., Hossain, M. M., Lucky, R. A., Bassi, A. S., & De Lasa, H. (2013). Integrated CO2 capture, wastewater treatment and biofuel production by microalgae culturing—A review. Renewable and Sustainable Energy Reviews, 27, 622–653. https://doi.org/10.1016/j.rser.2013.05.063
- Stefanidis, S. D., Kalogiannis, K. G., Iliopoulou, E. F., Michailof, C. M., Pilavachi, P. A., & Lappas, A. A. (2014). A study of lignocellulosic biomass pyrolysis via the pyrolysis of cellulose, hemicellulose and lignin. Journal of Analytical and Applied Pyrolysis, 105, 143–150. https://doi.org/10.1016/j.jaap.2013.10.013
- Alzahrani, A. M., & Rajendran, P. (2019). Petroleum Hydrocarbon and Living Organisms. IntechOpen. https://doi.org/10.5772/intechopen.86948
- Combustion | Definition, Reaction, Analysis, & Facts | Britannica. (2023, March 25). https://www.britannica.com/science/combustion
- Mlonka-Mędrala, A., Evangelopoulos, P., Sieradzka, M., Zajemska, M., & Magdziarz, A. (2021). Pyrolysis of agricultural waste biomass towards production of gas fuel and high-quality char: Experimental and numerical investigations. Fuel, 296, 120611. https://doi.org/10.1016/j.fuel.2021.120611
- Sun, X.-M., Ren, L.-J., Zhao, Q.-Y., Ji, X.-J., & Huang, H. (2018). Microalgae for the production of lipid and carotenoids: a review with focus on stress regulation and adaptation. Biotechnology for Biofuels, 11(1), 272. https://doi.org/10.1186/s13068-018-1275-9
- Wen, Z., & Johnson, M. B. (2009). Microalgae as a feedstock for biofuel production. Virginia Cooperative Extension, Publication 442-886.
- Firdaus, M. Y., Guo, Z., & Fedosov, S. N. (2016). Development of kinetic model for biodiesel production using liquid lipase as a biocatalyst, esterification step. Biochemical Engineering Journal, 105, 52–61. https://doi.org/10.1016/j.bej.2015.09.003
- Biofuels Basics. (n.d.). Retrieved April 1, 2023, from https://www.nrel.gov/research/re-biofuels.html
- Bhatia, S. C. (2014). Advanced Renewable Energy Systems, (Part 1 and 2). India: WPI India.
- Melis, A. (2009). Solar energy conversion efficiencies in photosynthesis: Minimizing the chlorophyll antennae to maximize efficiency. Plant Science, 177(4), 272–280. https://doi.org/10.1016/j.plantsci.2009.06.005
- Alishah Aratboni, H., Rafiei, N., Garcia-Granados, R., Alemzadeh, A., & Morones-Ramírez, J. R. (2019). Biomass and lipid induction strategies in microalgae for biofuel production and other applications. Microbial Cell Factories, 18(1), 178. https://doi.org/10.1186/s12934-019-1228-4
- Pandey, M. K., Dasgupta, C. N., Mishra, S., Srivastava, M., Gupta, V. K., Suseela, M. R., & Ramteke, P. W. (2019). Bioprospecting microalgae from natural algal bloom for sustainable biomass and biodiesel production. Applied Microbiology and Biotechnology, 103(13), 5447–5458. https://doi.org/10.1007/s00253-019-09856-2
Image References
- Banner: CSIRO Science Image – CSIRO Science Image. (n.d.). Retrieved April 13, 2023, from https://www.scienceimage.csiro.au/image/10697/
- https://euglenanetwork.org/2022/11/22/sequencing-project-to-unleash-the-biotechnology-potential-of-euglenoids/
- Figure 1: (Harmful Algal Bloom in San Francisco Bay Results in Aquatic Mortality, Fish Kills – California Ocean Protection Council, n.d.)
- Figure 2: Firdaus, M. Y., Guo, Z., & Fedosov, S. N. (2016). Development of kinetic model for biodiesel production using liquid lipase as a biocatalyst, esterification step. Biochemical Engineering Journal, 105, 52–61. https://doi.org/10.1016/j.bej.2015.09.003
- Figure 3: Pond-cultivated algae: Slimy superhero for aquafeeds? – Responsible Seafood Advocate. (2019, May 27). Global Seafood Alliance. https://www.globalseafood.org/advocate/pond-cultivated-algae-slimy-superhero-aquafeeds/
- Figure 4: Ooms, M. D., Dinh, C. T., Sargent, E. H., & Sinton, D. (2016). Photon management for augmented photosynthesis. Nature Communications, 7(1), 12699. https://doi.org/10.1038/ncomms12699